DEVELOPMENT – Streamline Formulation
and Stop Wasting Time
Reduce Formulation Time & Costs
Avoid missing changes made to marketing briefs and keep your product development team on the same page. Prevent costly development errors that cause re-formulation, re-testing and re-labeling.
Fast Track Development
Find ingredients to use in your formulations and review regulatory and safety data on any ingredient. Check formulas against Retailer Clean Lists.
Step-by-Step Guidance
Follow our guided process to qualify and approve new Raw Materials for your formulations.
Find Ingredients, Build Compliant Products, Access Documents Easily
The IIN is the only ingredient, regulatory, and document management software, which enables you to create compliant products from the start.
Track formula version history and any changes made
Ithos’ industry-leading formulation PLM makes it easy to ensure each product is compliant with design and manufacturing regulations before it goes to market. Achieve an additional level of product compliance by checking formulas against internal or leading retailer Clean Lists.
All reformulation history is tracked, so your team can view past formula iterations, what red flags were raised, and how compliance issues were solved.

Quickly find ingredients to use or replace in a formula
The IIN removes your need to search multiple websites and complicated databases. Using the Ingredient Search module, you can find ingredients to use in formulas, find Raw Material and ingredient replacements and discover details on any ingredient by searching its INCI or SKU.

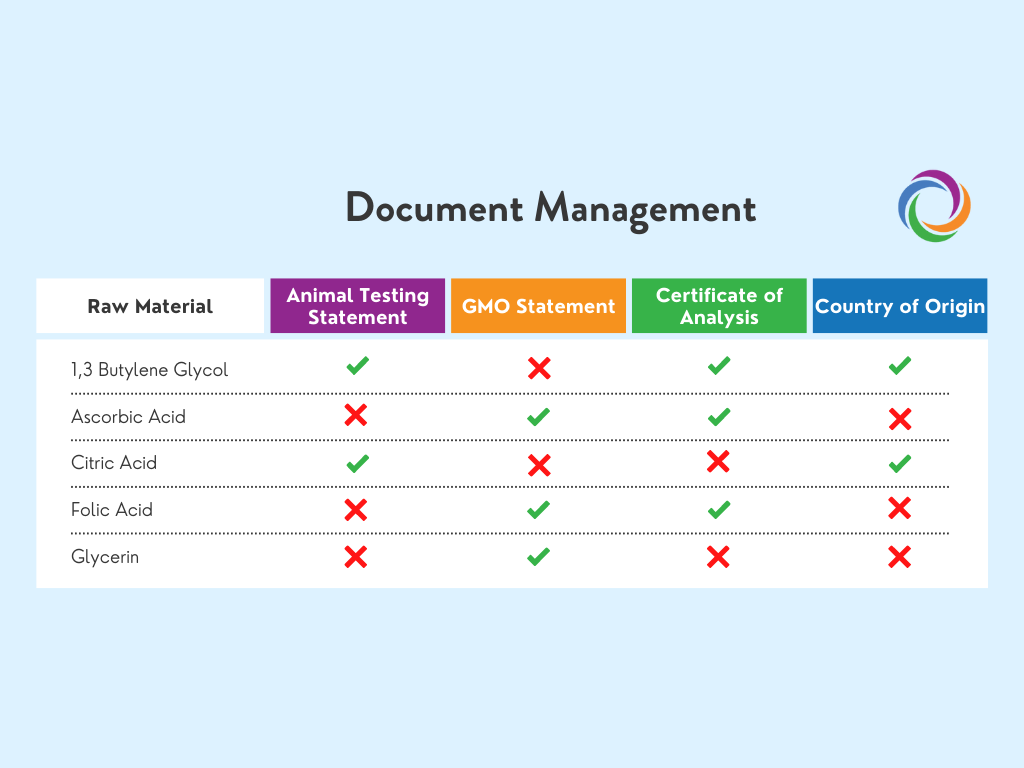
Access all documentation in one place
Qualify/Validate ingredients before you use them. Set standard requirements for raw materials before you use them. Make sure everyone who needs access to documentation has it, and put controls in place to maintain the quality of your information.
What Stage are you at in New Product Development?
NPD Wheel
Click around the spokes of the Ithos NPD Wheel to learn more about each stage!
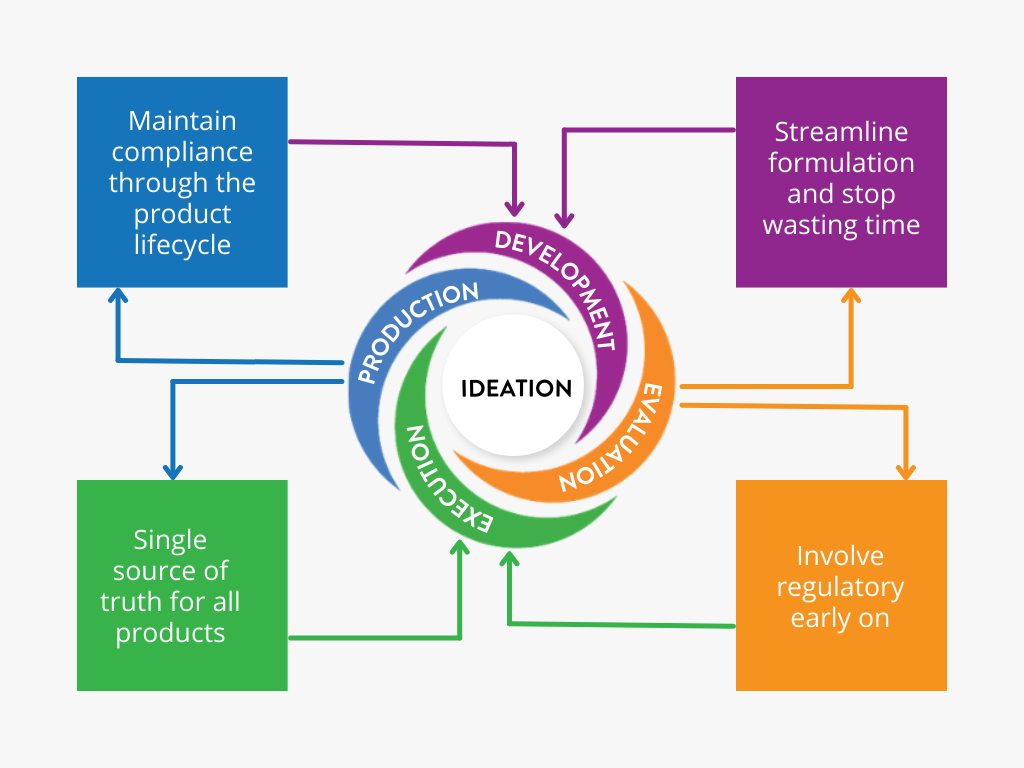
Ideation
Is it difficult to get everyone on the same page for new product development?
Do you receive incomplete marketing briefs? Or miss changes made to it after the fact?
Development
Do you have trouble tracking the various tests for formulas or knowing what testing is needed?
How do you track the reasons for reformulation?
Need to find alternative ingredients?
Evaluation
Do you have trouble gathering missing documents for your RMs?
Does it take you too long to review formulas for global compliance?
Is it hard for you to stay on top of the changing ingredient laws?
Execution
Are you missing documents needed for product registrations?
Does it take a lot of time for you to gather missing documents?
Do you need SDSs quickly, last minute?
Production
Do you find yourself facing the costs associated with noncompliance due to an ingredient ban or usage restriction?
Have you experienced difficulty maintaining compliance with changing regulations that require additional reporting?
Is it hard to find where an ingredient is used across your product portfolio?
Do you often miss expired docs until you need them?
Is it difficult to track finished good imports and the required data points?
Development
Do you have trouble tracking the various tests for formulas or knowing what testing is needed?
How do you track the reasons for reformulation?
Need to find alternative ingredients?
Evaluation
Do you have trouble gathering missing documents for your RMs?
Does it take you too long to review formulas for global compliance?
Is it hard for you to stay on top of the changing ingredient laws?
Execution
Are you missing documents needed for product registrations?
Does it take a lot of time for you to gather missing documents?
Do you need SDSs quickly, last minute?
Production
Do you find yourself facing the costs associated with noncompliance due to an ingredient ban or usage restriction?
Have you experienced difficulty maintaining compliance with changing regulations that require additional reporting?
Is it hard to find where an ingredient is used across your product portfolio?
Do you often miss expired docs until you need them?
Is it difficult to track finished good imports and the required data points?
Trusted by Leading Cosmetics Organizations
Subscribe to Our Newsletter
Sign up to receive updates from Ithos Global, including information about upcoming events, regulatory news, and more.
Ithos Global Names John Bender as New President to Lead Growth and Expansion
Last Updated on July 11, 2024 by Ithos Global Regulatory Team Experienced SaaS Industry Executive to Drive Growth and Operational…
3 Reasons to Come See Us at NYSCC Suppliers’ Day 2024!
Last Updated on April 16, 2024 by Ithos Global Regulatory Team We‘re heading back to the Big Apple May 1-2…
Debunking MoCRA Myths
MoCRA Myths Debunked: Reporting Tips to Save You Time Like anything new, there’s some misinformation floating around about MoCRA. And…
8 Things You Can Do Now to Prepare for MoCRA Webinar
SUMMARY The Modernization of Cosmetics Regulation Act (MoCRA) was signed into law as part of the 2023 Omnibus Bill. It…
EC#
EU-specific, seven digit substance registration number with pattern xxx-xxx-x, either Einecs, Elinecs, No-Longer Polymers databases.
IL
“Ingredient Listing” or “List of Ingredients” or ‘Cosmetic Ingredient Declaration”, listing of all intentionally added ingredients (including substances and raw…
FDA
US Food and Drug Administration (a US federal regulator)
Wednesday, May 29th ‘Debunking MoCRA Myths’ Webinar Signup
MoCRA Myths Debunked: Reporting Tips to Save You Time Wednesday, May 29th 12pm – 1pm EST Since the Modernization…
May 2024 MoCRA Webinar Signup
Join us for two free webinars! Our regulatory team is guiding brands, manufacturers, retailers and labs on MoCRA compliance. Now,…